Courtney Gray Montgomery, Ph.D.
Professor
Genes & Human Disease Research Program
My 101
Genes determine your height, hair color and even, in some cases, whether or not you will get certain diseases. Much of what scientists know about human disease today has been learned from the study of genetics, and in my lab we focus on how genes can trigger the onset of disease, make disease worse or even why they express themselves differently in one person versus another. In particular, we study the roles genes play in auto-immune and auto-inflammatory diseases.
While I began my career in the field of cancer genetics, I never lost ties to the work I began as a graduate student in genetics of the immune system. I now direct the Sarcoidosis Research Unit and lead projects focused on understanding the genetic and environmental risk factors of sarcoidosis. This “medical mystery” occurs when small nodules called granulomas form in and around organs. Our goal is to better understand the risk factors of sarcoidosis so that we can better diagnose, treat and even prevent disease.
Research
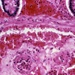
My laboratory is focused on the identification of genes predisposing to complex diseases, particularly sarcoidosis, an inflammatory disorder that can affect any organ in the body. It is characterized by growths called granulomas, much like those found in people with Tuberculosis. Patients can have granulomas in the liver, lymph glands, bone marrow, even the brain, but are most frequently diagnosed because of granulomas in the lungs. The disease can resolve on its own or can be chronic, leading to severe health problems. We know that sarcoidosis can run in families, but we also know that certain environmental exposures increase the risk of disease in certain people with a particular genetic background. For example, sarcoidosis is more prevalent in women and, in the United States, African Americans are both more commonly and more severely affected than Caucasians.
It is the goal of my laboratory to not only find the genes that make someone susceptible to disease but also to understand why the disease is worse in some patients compared to others. Specifically, our studies have led the way in the genetics of sarcoidosis for over a decade and now focus on understanding how genes cause particular cells within the immune system to respond in such a way to make granulomas form.
We are so thankful to the patients that participate in our research clinics and share not only their time and participate in our studies, but also share their amazing stories with us!
Brief CV
Education
B.A. (honors), Oklahoma City University, 1995
M.S., University of Oklahoma Health Sciences Center, 2000
Ph.D., Case Western Reserve University, Cleveland, Ohio, 2004
Honors and Awards
Outstanding Science Student Award, Oklahoma City University, 1995
Rhodes Scholar Semifinalist, Oxford University, 1995
2nd Place, College of Public Health Graduate Student Research Competition, University of Oklahoma Health Sciences Center, 1999
Graduate Student Association Award, University of Oklahoma Health Sciences Center, 2000
NHLBI trainee fellow, Case Western Reserve University Division of Genetic and Molecular Epidemiology, 2000-2002
Nominee for C.W. Cotterman Award, American Society of Human Genetics, 2001
Student of the Year Award, Case Western Reserve University Division of Genetic and Molecular Epidemiology, 2002-2003
J. Donald and Patricia Capra Award for Scientific Achievement, 2012
Other Activities
Editor, BMC Genetics, Genetic Analysis Workshop, 2004-2005, 2007
Reviewer, Human Heredity, 2005-present
Reviewer, Genes and Immunity, 2005-present
Reviewer, BioTechniques, 2005-present
Reviewer, The Journal of Clinical Endocrinology & Metabolism, 2006-present
Editorial Board Member, Open Genetics Journal, 2007-present
Reviewer, Annals of Human Genetics, 2007-present
Reviewer, Biometrical Journal, 2007-present
Memberships
Rare Disease Consortia for Neurosarcoidosis
Trans-omics for Precision Medicine (TOPMed) Consortia, NHLBI, NIH
American Association of Sarcoidosis and Other Granulomatous Diseases - Executive Committee
American College of Rheumatology
American Thoracic Society
American Society of Human Genetics
International Genetic Epidemiology Society
Joined OMRF scientific staff in 2008
Publications
Recent Publications
Myers JM, Sandel C, Alvarez K, Garman L, Wiley G, Montgomery C, Gaffney P, Stavrakis S, Fairweather D, Bruno KA, Zhao YD, Cooper LT, Cunningham MW. Cardiac autoantibodies promote a fibrotic transcriptome and reduced ventricular recovery in human myocarditis. Front Immunol 16:1500909, 2025 March, PMID: 40181955, PMCID: PMC11965655
Li R, Taliun SAG, Liao K, Flickinger M, Sobell JL, Genovese G, Locke AE, Chiu RR, LeFaive J, Martins T, Chapman S, Neumann A, Handsaker RE, Arnett DK, Barnes KC, Boerwinkle E, Braff D, Cade BE, Fornage M, Gibbs RA, Hoth KF, Hou L, Kooperberg C, Loos RJF, Metcalf GA, Montgomery CG, Morrison AC, Qin ZS, Redline S, Reiner AP, Rich SS, Rotter JI, Taylor KD, Viaud-Martinez KA, NHLBI Trans-Omics for Precision Medicine (TOPMed) Consortium, Genomic Psychiatry Cohort investigators, Bigdeli TB, Gabriel S, Zollner S, Smith AV, Abecasis G, McCarroll S, Pato MT, Pato CN, Boehnke M, Knowles J, Kang HM, Ophoff RA, Ernst J, Scott LJ. Whole genome sequence-based association analysis of African American individuals with bipolar disorder and schizophrenia. medRxiv, 2024 December, PMID: 39763555, PMCID: PMC11703280
Keener R, Chhetri SB, Connelly CJ, Taub MA, Conomos MP, Weinstock J, Ni B, Strober B, Aslibekyan S, Auer PL, Barwick L, Becker LC, Blangero J, Bleecker ER, Brody JA, Cade BE, Celedon JC, Chang YC, Cupples LA, Custer B, Freedman BI, Gladwin MT, Heckbert SR, Hou L, Irvin MR, Isasi CR, Johnsen JM, Kenny EE, Kooperberg C, Minster RL, Naseri T, Viali S, Nekhai S, Pankratz N, Peyser PA, Taylor KD, Telen MJ, Wu B, Yanek LR, Yang IV, Albert C, Arnett DK, Ashley-Koch AE, Barnes KC, Bis JC, Blackwell TW, Boerwinkle E, Burchard EG, Carson AP, Chen Z, Chen YI, Darbar D, de Andrade M, Ellinor PT, Fornage M, Gelb BD, Gilliland FD, He J, Islam T, Kaab S, Kardia SLR, Kelly S, Konkle BA, Kumar R, Loos RJF, Martinez FD, McGarvey ST, Meyers DA, Mitchell BD, Montgomery CG, North KE, Palmer ND, Peralta JM, Raby BA, Redline S, Rich SS, Roden D, Rotter JI, Ruczinski I, Schwartz D, Sciurba F, Shoemaker MB, Silverman EK, Sinner MF, Smith NL, Smith AV, Tiwari HK, Vasan RS, Weiss ST, Williams LK, Zhang Y, Ziv E, Raffield LM, Reiner AP, NHLBI Trans-Omics for Precision Medicine (TOPMed) Consortium, TOPMed Hematology and Hemostasis Working Group, TOPMed Structural Variation Working Group, Arvanitis M, Greider CW, Mathias RA, Battle A. Validation of human telomere length multi-ancestry meta-analysis association signals identifies POP5 and KBTBD6 as human telomere length regulation genes. Nat Commun 15:4417, 2024 May, PMID: 38789417, PMCID: PMC11126610
Selected Publications
Garman L, Pelikan RC, Rasmussen A, Lareau CA, Savoy KA, Deshmukh US, Bagavant H, Levin AM, Daouk S, Drake WP, Montgomery CG. Single cell transcriptomics implicate novel T cell and monocyte immune dysregulation in sarcoidosis. Frontiers in Immunology. December 8 2020; 11:567342. doi: 10.3389/fimmu.2020.567342. eCollection 2020. PMID 33363531. PMCID: PMC7753017
Taliun G, …Montgmery C, …, Abecasis G. Sequencing of 53,831 diverse genomes from the NHLBI TOPMed Program. Nature. February 11, 2021;590(7845):290-299. Doi: 10.1038/s41586-021-03205-y. Epub 2021 Feb 10. PMID: 33568819, PMCID: PMC7875770
Pelikan R, Kelly JA, Fu Y, Laraeu CA, Tessneer KL, Wiley GB, Wiley M, Glenn SB, HarleyJB, Guthridge JM, James JA, Aryee MJ, Montgomery CG, Gaffney PM. Enhancer histone-QTLs are enriched on autoimmune disease risk haplotypes and influence gene expression variability within chromatin networks. Nature Communications, 2018. PMID: 30046115 PMCID: 6060153
Lareau CA, DeWeese CF, Adrianto I, Lessard CJ, Gaffney PM, Iannuzzi MC, Rybicki BA, Levin AM, Montgomery CG. Polygenic Risk Assessment Reveals Pleiotropy between Sarcoidosis and Inflammatory Disorders in the Context of Genetic ancestry. Genes Immun. 2017 Mar;18(2):88-94. PMID: 28275240, PMCID: PMC5407914
Bello GA, Adrianto I, Dumancas GD, Levin AM, Innauzzi MC, Rybicki BA, Montgomery C. Role of NOD2 pathway genes in sarcoidosis cases with clinical characteristics of Blau Syndrome. Am J of Resp Crit Care Med. 2015 Nov 1;192(9):1133-5. PMID: 26517420 PMCID: 5447311
Adrianto I, Lin CP, Hale JJ, Levin AM, Datta I, Parker R, Adler A, Kelly JA, Kaufman KM, Lessard CJ, Moser KL, Kimberly RP, Harley JB, Iannuzzi MC, Rybicki BA, Montgomery CG†. Genome-wide association study of African and European Americans implicates multiple shared and ethnic specific loci in sarcoidosis susceptibility. PLoS One. 2012;7(8):e43907. PMID: 22952805 PMCID: 3428296
Contact
Genes & Human Disease Research Program, MS 57
Oklahoma Medical Research Foundation
825 N.E. 13th Street
Oklahoma City, OK 73104
Phone: (405) 271-2468
Fax: (405) 271-2578
E-mail: Courtney-Montgomery@omrf.org
For media inquiries, please contact OMRF’s Office of Public Affairs at news@omrf.org.
Lab Staff
Astrid Rasmussen, Ph.D.
Research Associate Professor
Christopher Dunn, Ph.D.
Postdoctoral Scientist
Caleb Watkins
Data Analyst
Kiely Grundal
Senior Manager of Laboratory
Judy Harris
Lead Clinical Research Nurse
Sharon Johnson
Research Data Coordinator
Jordan Odell Brown
Graduate Student
Melanie Jones
Administrative Assistant II
Carly McHenry
Administrative Assistant
News from the Montgomery lab

OMRF’s education programs enjoyed a record summer in 2022. For most students, summer means late nights, lake days and snooze buttons. But for those who spend the dog days in OMRF’s labs, it’s mice, pipettes and data analysis. Despite the obvious appeal of a lazy few months between semesters, in recent years, OMRF has seen […]

The National Institutes of Health has awarded 17 grants worth a total of $14.7 million to OMRF. The grants are part of the $10 billion in economic stimulus funds that will be provided for medical research through the American Recovery and Reinvestment Act of 2009. The grants will fund OMRF research on a wide array […]
The National Institutes of Health has awarded two grants worth a total of $26.3 million to OMRF for research into anthrax and to help train new scientists. Each grant will allow scientists to continue research started in 2004 and 2005 and keep them working through 2014 on several interconnected projects. In the first project, a $14.5 […]
A new wave of researchers has joined the Oklahoma Medical Research Foundation’s scientific staff as part of the foundation’s expansion. OMRF has added seven new scientists to its staff. In addition, two research assistants have been promoted to faculty-level positions. The new researchers have come to OMRF from a variety of institutions across the U.S. […]








































