Each week, OMRF Vice President of Research Dr. Rod McEver opens “Adam’s Journal” to answer a medical question from Adam Cohen, OMRF’s senior vice president & general counsel and interim president.
Adam’s Journal
I’ve read about monoclonal antibodies as a treatment for Covid-19. What are they, and are they alternatives to vaccination?
Dr. McEver Prescribes
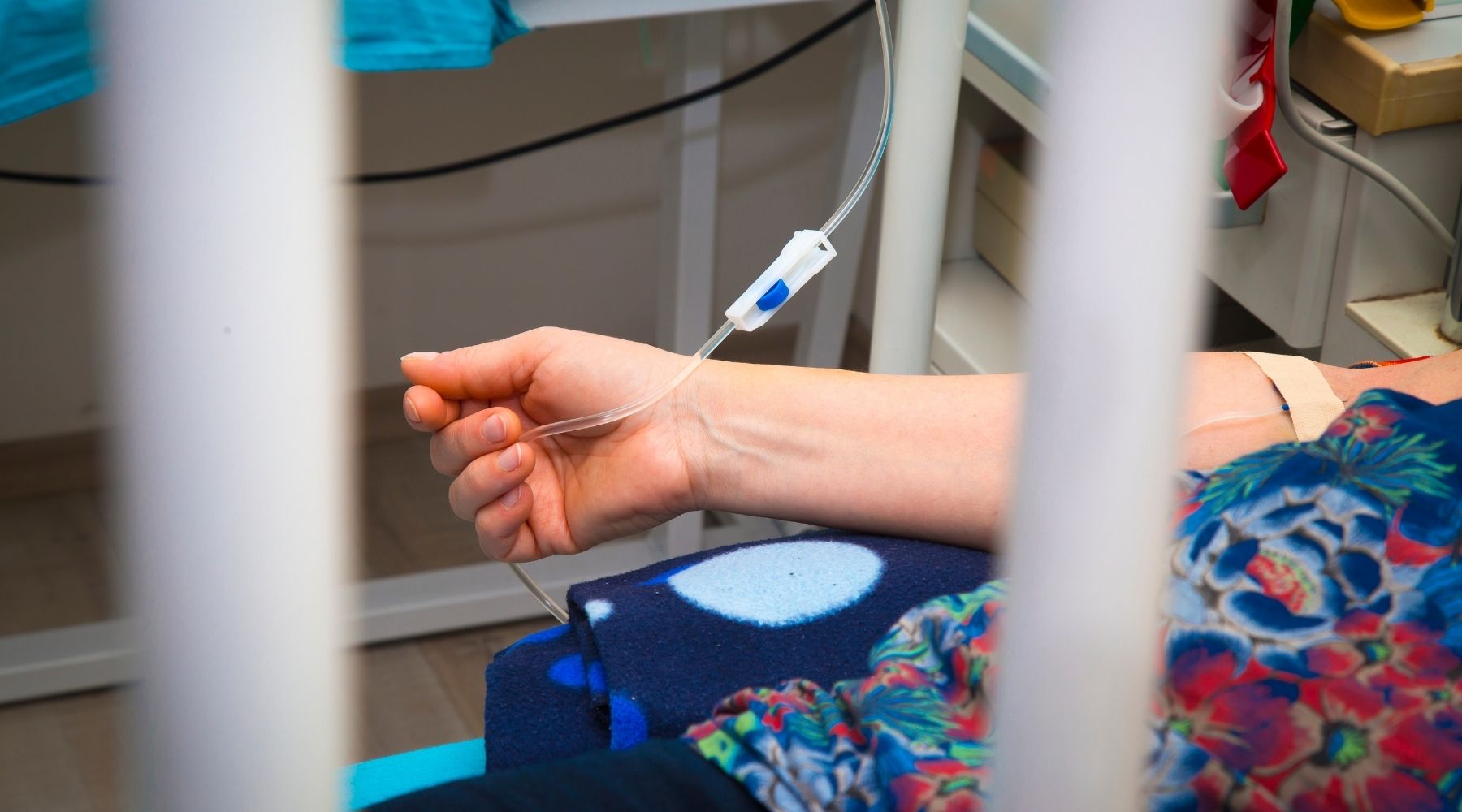
Monoclonal antibodies are lab-made proteins designed to fight viruses and other conditions. While they can lessen the symptoms of the virus if administered prior to serious illness (and, in some cases, after exposure to the virus), they aren’t substitutes for vaccination.
The U.S. Food and Drug Administration has granted emergency use authorization for three monoclonal antibody treatments for Covid-19. Health care providers administer them by intravenous infusion or injection.
The FDA has okayed the treatments for those who test positive for Covid-19, have had symptoms for 10 days or less, and are at high risk for severe disease. It has also okayed the use of one, produced by Regeneron, as a preventive therapy for certain high-risk people who’ve been exposed to someone with Covid-19 (or who are likely to be exposed, such as nursing home residents).
In clinical trials, Regeneron’s therapy reduced hospitalization and death in high-risk Covid-19 patients by 71%. In another trial, it reduced the risk of contracting Covid-19 by 81% in people with normal immune systems.
Early intervention is key for someone with Covid-19 or exposed to the virus to benefit from monoclonal antibodies. Treatment should start as soon as possible after a positive test or exposure.
While monoclonal antibody treatments for Covid-19 are new, monoclonal antibodies are not. At the Oklahoma Medical Research Foundation, our scientists have studied and produced them for years as research tools and as potential therapies. Soliris and Adakveo, two of three FDA-approved drugs born from OMRF discoveries, are monoclonal antibodies.
–
Do you have a health query for Dr. McEver? Email contact@omrf.org and your question may be answered in a future column!