Gabriel Pardo, M.D.
Director, OMRF Multiple Sclerosis Center of Excellence
Professor
Arthritis & Clinical Immunology Research Program
My 101
The focus of OMRF’s Multiple Sclerosis Center of Excellence is to provide optimal medical care while advancing the understanding of MS—a disease of the central nervous system. Multiple sclerosis is an autoimmune disease of the brain and spinal cord caused by inflammation and degeneration. Inflammation causes damage to myelin, the protective covering that surrounds nerve cells. When this covering is damaged, nerve impulses are slowed and sometimes totally blocked. Degeneration can cause nerve cells to die prematurely. This combination of effects causes a variety of symptoms, including problems with vision, tremors, paralysis, painful spasms, imbalance, and cognitive changes.
From an MS clinical research perspective, we are interested in the visual system, cognition, balance and ambulation in MS.
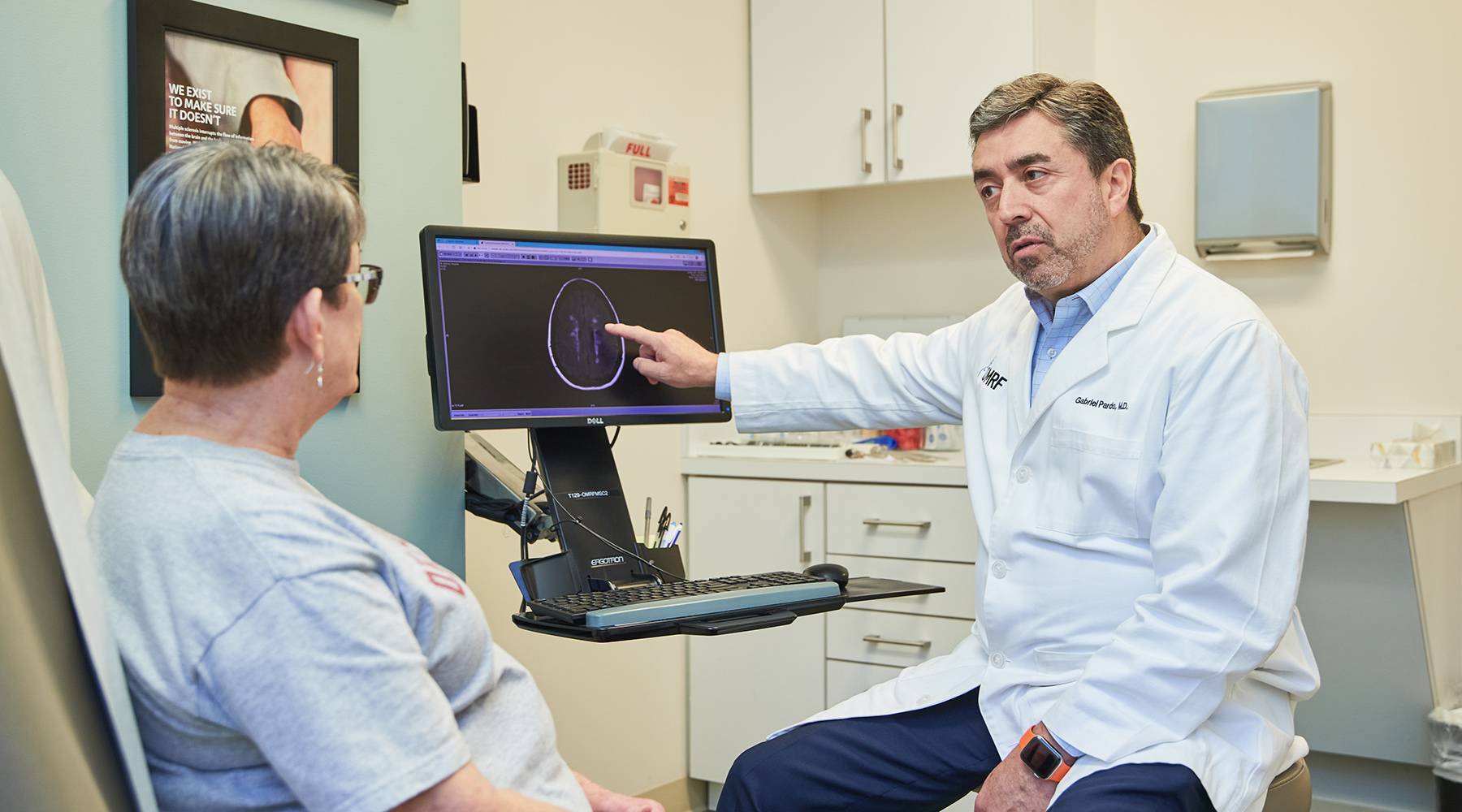
The effects of MS in the brain commonly involve information processing in the visual system. The field of neuro-ophthalmology deals with problems with vision such as information coming from the eyes to the brain (called the afferent system) and the brain control of eyes positioning and movement (called the efferent system). Damage to the nerve pathways in the afferent system causes decrease or loss of vision, or blind spots in the visual fields. Damage to the efferent system creates misalignment of the eyes causes double vision or tracking abnormalities. Through clinical trials, we hope to determine if evaluating certain measurements in the eye can give us insight into degenerative processes in the brain and use those measurements as markers of disease progression.
One area of research we are pioneering is the effect of MS on balance. The delay of messages from the brain to the body can cause overcorrection causing patients to fall. It can alter normal gait, making it impossible to walk independently. We want to design objective ways to monitor changes and how the disease progression can impact patients’ function and safety. This will help identify the problems in a timely manner and assist us in intervening accordingly.
In addition to working with patients to address MS-related motor skills difficulties, sensory issues, cognition, and coordination, the Center participates in clinical trials to evaluate new medications directed at treating the disease.
We also work with scientists in the lab exploring basic science aspects of MS such as genetics and immunology. By learning more about the origins and behaviors of the disease, we hope to be able to slow or stop progression in current patients and prevent MS in others.
Research
Multiple sclerosis poses many scientific challenges that cover the spectrum from molecular research to clinical outcomes. A hindrance to all type of research and clinical interventions is the relative lack of objective quantifiable measures of disease progression. These “disease markers” are yet to be identified and validated and may include autoantibodies, cytokines, MRI parameters, anatomical neuronal evaluation, and disability measures.
A clinical research interest of our center is the use of computerized systems to evaluate gait and balance. This approach identifies changes before they are apparent on regular physical examinations and can be used to accurately monitor progression. Another is the use of optical coherence tomography to measure potential neuronal loss in the retina as an indicator of a corresponding process in the brain that is secondary to MS-induced inflammation and/or neurodegeneration.
Brief CV
Education
B.S., Colegio San Carlos, Bogotá, Colombia, 1980
M.D. Universidad Militar Nueva Granada, Bogotá, Colombia, 1985
Ophthalmology Residency, Universidad Militar Nueva Granada, Bogotá, Colombia
Neuro-Ophthalmology Fellowship, University of Texas, Galveston, TX
Neurology Residency, University of Oklahoma, Oklahoma City, OK
Honors and Awards
Hospital Militar Central Resident’s Best Research Project: “Chlamydial Iga Antibodies In Tears Of Humans With Follicular Conjunctivitis”, 1990
American Medical Association, Physician’s Recognition Award, 2000
Volunteer Hall of Fame, Health Professionals / Research category, NMSS, 2008
Honorary Chairman, MS Walk OKC, NMSS, 2010
Labe Scheinberg Award in Neurorehabilitation, Consortium of Multiple Sclerosis Centers, 2017
Hope Award in Outstanding Community and Humanitarian Services, NMSS, 2018
Other Activities
Member, Board of Trustees, National Multiple Sclerosis Society, Oklahoma Chapter, 2001-present
Board Member, MS Bridge, 2003-present
Vice Chairman, Clinical Action Committee, National Multiple Sclerosis Society, Oklahoma Chapter, 2005-2006
Chairman, Clinical Action Committee, National Multiple Sclerosis Society, Oklahoma Chapter, 2007-present
Member, National Council of Clinical Action Committee Chairs, National Clinical Advisory Board, National Multiple Sclerosis Society, 2008-present
Reviewer, International Journal of MS Care, 2006-present
Reviewer, Neurology, 2000-present
Reviewer, Ocular Therapeutics Handbook – Sudden Visual Loss chapter, Editor: Nicky R. Holdeman, 1995
Memberships
American Academy of Neurology
American Academy of Ophthalmology
American Medical Association
North American Neuro-Ophthalmology Society
Multiple Sclerosis Section
Neuro-Ophthalmology / Neuro-Otology Section AAN
Oklahoma State Medical Association
Oklahoma County Medical Society
Joined OMRF scientific staff in 2011
Publications
Recent Publications
Cerqueira JJ, Berthele A, Cree BAC, Filippi M, Pardo G, Pearson OR, Traboulsee A, Ziemssen T, Vollmer T, Bernasconi C, Mandel CR, Kulyk I, Chognot C, Raposo C, Schneble HM, Thanei GA, Incera E, Havrdová EK. Long-Term Treatment With Ocrelizumab in Patients With Early-Stage Relapsing MS: Nine-Year Data From the OPERA Studies Open-Label Extension. Neurology 104:e210142, 2025 February, PMID: 39883906, PMCID: PMC11781784
Cordeiro B, Ahn JJ, Gawde S, Ucciferri C, Alvarez-Sanchez N, Revelo XS, Stickle N, Massey K, Brooks DG, Guthridge JM, Pardo G, Winer DA, Axtell RC, Dunn SE. Obesity intensifies sex-specific interferon signaling to selectively worsen central nervous system autoimmunity in females. Cell Metab, 2024 August, PMID: 39168127, PMCID: PMC11463735
Anderson E, Powell M, Yang E, Kar A, Leung TM, Sison C, Steinberg R, Mims R, Choudhury A, Espinosa C, Zelmanovich J, Okoye NC, Choi EJ, Marder G, Narain S, Gregersen PK, Mackay M, Diamond B, Levy T, Zanos TP, Khosroshahi A, Sanz I, Luning Prak ET, Bar-Or A, Merrill J, Arriens C, Pardo G, Guthridge J, James J, Payne A, Utz PJ, Boss JM, Aranow C, Davidson A. Factors associated with immune responses to SARS-CoV-2 vaccination in autoimmune disease individuals. JCI Insight, 2024 June, PMID: 38833310, PMCID: PMC11383356
Selected Publications
Fjeldstad-Pardo C, Thiessen A, Pardo G. Telerehabilitation in multiple sclerosis: Results of a randomized feasibility and efficacy pilot study. International Journal of Telerehabilitation. 10(2): 55-64. Fall 2018. PMID: 30588276 PMCID: PMC6296802
Pardo G, Jones D. The sequence of disease-modifying therapies in relapsing multiple sclerosis: safety and immunologic considerations. Journal of Neurology. 264(12): 2351-2374. December 2017 (Epub September 6, 2017). PMID: 28879412 PMCID: PMC5688209
McCroskery P, Selmaj K, Fernandez O, Grimaldi LM, Silber E, Pardo G, Freedman M, Zhang Y, Xu L, Cadavid D, Mellion M. Safety and tolerability of opicinumab in relapsing multiple sclerosis: the Phase 2b SYNERGY Trial. Neurology Apr 2017, 88 (16 Supplement) P5369. PMID: 31285147
Schubert RD, Hu Y, Kumar G, Szeto S, Abraham P, Winderl J, Guthridge JM, Pardo G, Dunn J, Steinman L, Axtell R. IFN-β treatment requires B cells for efficacy in neuroautoimmunity. The Journal of Immunology. 194(5): 2110-2116. March 2015. PMID: 25646307 PMCID: PMC4340715
Fjeldstad C, Fjeldstad A, Weir J, Pardo G. Association of vitamin D deficiency with RNFL thickness in MS individuals without history of optic neuritis. Multiple Sclerosis and Related Disorders. 3(4): 489-493. 2014. PMID: 25877061
Fjeldstad C, Bemben M, Pardo G. Reduced retinal nerve fiber layer and macular thickness in patients with multiple sclerosis with no history of optic neuritis identified by the use of spectral domain high-definition optical coherence tomography. Journal of Clinical Neuroscience. 18(11): 1469-1472. November 2011. PMID: 21917458
Contact
Multiple Sclerosis Center of Excellence
Arthritis & Clinical Immunology Research Program, MS 50
Oklahoma Medical Research Foundation
825 N.E. 13th Street
Oklahoma City, OK 73104
Phone: (405) 271-6242
Fax: (405) 271-2887
E-mail: Gabriel-Pardo@omrf.org
For media inquiries, please contact OMRF’s Office of Public Affairs at news@omrf.org.
Clinic Staff
Chelsea Berkley, M.D.
Clinical Assistant Member
Cecilie Fjeldstad Pardo, Ph.D.
Clinical Research Scientist
Anthony "Tony" Sharp
Physician Assistant
Jennifer Smith
Physician Assistant
Misty Mihelich
Clinic Manager
Donna Prickett
RN Case Manager
Michelle "Micki" Drake
Clinical Research Coordinator
Kellie Kraus
Clinical Research Coordinator
Lauren Still, LCSW
Clinical Social Worker
Charlotte Latham
Phlebotomist/Medical Assistant
Tracey Nail
Medical Assistant
Berlinda Thompson
Medical Assistant
Brittnee Wilson
Patient Access Representative
Janice Woodson
Receptionist Secretary
Carmen Childs
Affiliate
Bobbette Miller
Affiliate
Lori Lowe
Administrative Assistant III
News from the Pardo lab
May Dr. Scott Plafker likes medical solutions that don’t come in pill bottles. In his OMRF lab, he’s studied how a compound naturally produced in broccoli, cabbage, cauliflower and Brussels sprouts can protect eyes as they age. “Because it’s readily available in vegetables, people can get it with ease, and they can get it without […]

Leah Campbell lost her sight to an unknown illness. Then it stole her ability to move. Could doctors stop the mystery disease before it took her life? Leah Campbell’s days begin about the time the sun rises. After waking and having breakfast, she answers dozens of emails. Often, she composes just as many new […]