Clinical Genomics Center
The OMRF Clinical Genomics Center (CGC) is the preeminent sequencing facility in Oklahoma. Equipped with an Illumina NovaSeq 6000, Illumina NextSeq 2000, Illumina MiSeq, Illumina iSeq, Oxford Nanopore PromethION 2, and Oxford Nanopore MinION, there are very few NGS projects the OMRF CGC is not capable of performing in a rapid, cost-effective manner. The center is additionally equipped with an Illumina iScan and Autoloader for extremely-high throughput genotyping projects.
The CGC seeks to support all aspects of the local biotech industry and academia. The center can perform all aspects of NGS, ranging from whole genome sequencing to single-cell transcriptomics. The CGC welcomes the opportunity for community outreach and has hosted many tours of its facilities as well as provided guest lecturers in various classes, departmental seminars, and industry-sponsored seminars.
Pricing and Submissions
Accessing Data
Upon completion of a run all read data will be converted into fastq format and placed on an accessible FTP site. Information on accessing this site will be emailed to the respective researcher as soon as it is available. The FTP site is only a temporary storage site, however, and all data is subject to deletion without notice after two weeks.
Bioinformatics and Training
Training is available through OMRF’s Center for Biomedical Data Sciences.
Contact

Graham Wiley, Ph.D.
Room E-313
825 NE 13th St.
Oklahoma City, OK 73104
Contact
Research/Clinical Accounts and Billing/Insurance Inquiries
To contact the OMRF Clinical Laboratory staff, please call 405-271-7771.
Federal accreditation regulations prohibit laboratory personnel from providing patient information or results over the phone or via email. Patients, please contact your health care provider for questions concerning your results.

Flow Cytometry
The Flow Cytometry and Cell Sorting Core Facility supports many OMRF investigators by providing training, analysis and multi-parameter, high speed cell sorting.
Calendar
The Flow Cytometry Core uses iLab for scheduling equipment. Your PI needs to have registered with iLab before lab members are able to schedule an appointment.
The FacsAria and MoFlo are cell sorters and can be scheduled two weeks in advance. Appointments can be made from the times of 10:00 am to 5:00 pm with an hour off for lunch. Users cannot edit or delete their times for the sorters and should contact the lab to make changes. Cancellations for the Aria and MoFlo should be made 24 hours in advance; otherwise, there is a 1 hour cancellation charge.
The FacsCelesta, LSRII and Aurora are cell analyzers and can be reserved one month in advance. Trained users who feel confident to use our analyzers after hours (after 5 PM on the weekdays and the whole weekend) may do so. Any user that signs up before 8:30 AM on the LSRII should turn the machine on early, since it requires at least 10 minutes for warmup. The Aurora should be allowed to warm-up for at least 30 minutes. Users can edit and delete their times for the analyzer calendars.
There is a Kiosk workstation for starting and stopping your time. The billable time will be based on the Kiosk.
Fees
Flow cytometry analysis –Aurora, FacsCelesta & LSRII
Non-OMRF users
- Operator-assisted: $75/hr
- Independent user: $40/hr
- Minimum charge is 15 minutes
OMRF users
- Operator-assisted: $50/hr
- Independent user: $30/hr
- Minimum charge is 15 minutes
Cell sorting – MoFlo, FACSAria
Non-OMRF users
- Set-up fee: $30
- Hourly rate: $120/hr
- Analysis: $75/hr
- Minimum charge is 1 hour
OMRF users
- Set-up fee: $25
- Hourly rate: $100/hr
- Analysis: $50/hr
- Minimum charge is 1 hour
Corporate users
- Flow Cytometry Analysis: $50/hr
- Flow Cyometry Sorting: $100/hr and Set-up fee: $30
Getting Started
How to use kiosk
Kiosk Instructions for our analyzers (LSRII, Celesta, Calibur):
Here are the steps to access the Kiosk interface and begin your session on any analyzer within our facility.
To start your session with an already existing reservation:
- Please navigate to the Kiosk Interface URL: https://my.ilabsolutions.com/service_centers/4780/equipment_kiosk/authenticate
- You will log into the Kiosk interface using the same credentials you use to log into the main iLab site for the core.
- Once logged in, you will see a list of your pre scheduled reservations in “My kiosk sessions” (If you have multiple sessions, there is a search box to help guide you to the correct one you wish to start)
- Find your session, and to the right you should have a green “start” button. Once you click start, you will see the details of your reservation as well as a timer in the upper right hand corner.
- To navigate back to your list of sessions, click in the drop down menu where you see your name. Click my reservations.
- You may log out while your session is in process. To log out, click the upper right hand side menu and select Log out. On the log out screen, you will see your list of Active sessions.
To end your session:
- Please navigate to the Kiosk Interface URL: https://my.ilabsolutions.com/service_centers/4780/equipment_kiosk/authenticate
- You will log into the Kiosk interface using the same credentials you use to log into the main iLab site for the core.
- Find your current reservation in the list under “My kiosk sessions” and click the blue Finish button.
- A pop-up box will appear, asking you to confirm your action. Click “Finish session” again. Your time on the instrument has been logged.
New user registration
To register for an account:
- Navigate to the core page: https://OMRF.ilab.agilent.com/service_center/show_external/4780
- In the upper-right-hand corner of the screen, click ‘Register’.
- You will be directed to an authentication page where you will need to enter your Oklahoma Medical Research Facility credentials
- Once you have entered your credentials, click the ‘Login’ button
- You will be directed to an iLab Registration page where you will need to select your PI/Lab and verify your contact information
- Once your registration has been submitted, your PI will receive a notification that you have requested membership to their lab in iLab. They will need to approve your membership and assign any OMRF Account Numbers for your use.
To Create an Equipment Reservation:
Once you have been accepted into your PI’s lab and assigned an account number (s), you can schedule equipment time.
- Navigate to the core page: https://OMRF.ilab.agilent.com/service_center/show_external/4780
- At the upper right-hand of the page, click ‘Sign In’.
- Enter your OMRF Credentials and password, and sign in.
- Select the Schedule Equipment tab and click on the ‘View Schedule’ button next to the instrument of interest. Click and drag on the time frame you would like to schedule as your reservation.
- A window will pop up that will allow you to verify your reservations details and provide payment information before saving the reservation.
- To schedule time on one of the two sorters, you will need to fill out the preapproval form for every appointment. There is a way to book multiple appointments for the same type of experiment to avoid filling it out multiple times for the same work.
To use the equipment:
Before starting to use one of the analyzers, you must login at our Kiosk. When you are finished, log out at the Kiosk. The logged time will determine the time of instrument use for billing. If you fail to use the Kiosk, the booked time on the schedule will be billed as reserved. So if you use less than your scheduled time, it will be imperative to use the Kiosk to avoid over-billing, unless you tell Jacob or Diana to change the billed time.
PI registration
To register for an account:
- Navigate to the core page: https://OMRF.ilab.agilent.com/service_center/show_external/4780
- In the upper-right-hand corner of the screen, click ‘Sign In’ and use your OMRF logon.
- You will be directed to an authentication page where you will need to enter your Oklahoma Medical Research Facility credentials
- Once you have entered your credentials, click the ‘Login’ button
- You will be directed to an iLab Registration page where you will need to select your PI/Lab and verify your contact information
Before your lab members may schedule time on our equipment, you must “accept” them as “members” of your group and assign account numbers that they should use for Flow Core charges. You will receive emails as your lab members register for access to iLab. The request email will have specific instructions on how to approve the request. We have pasted instructions below.
Instructions for approving lab members to use iLab:
- Click here to log in: https://OMRF.ilab.agilent.com/service_center/show_external/4780
- Use your OMRF credentials to log into iLab. Then, look for the link in the left-hand menu that says ‘my labs’. Hover over and select your lab.
- Set the auto-approval amount if you do not wish to approve service requests below a certain dollar amount. To do this, select the ‘Members’ panel and enter a dollar amount in the ‘Auto Pre-Approval’ amount and click ‘save settings.’
- To approve lab membership requests, select the ‘Membership Requests & Account Number’ tab. New membership requests will show at the top of this page. Click “Approve” to accept a member into your lab. Click “Reject” if they are not a member of your lab.
- To assign an Account Number to a member of your lab, find the member in the above list where it says, ‘Manage Account Number.’ Select the checkbox(s) to the right of their name for the Account Number(s) you wish to assign them.
Additional resources
EasyPanel: Panel design and validation software
Learning about Cytometry and Diva software
Fluorescence Spectrum viewers
- BD Spectra Viewer
- BioLegend Spectra Viewer
- ThermoFisher Spectra Viewer
- Purdue University Cytometry Laboratories
- Scripps Research Flow Cytometry
- BD Biosciences
Instrumentation
The Beckman Coulter Moflo XDP1 sorter purchased in 1998 is equipped with a Saphhire 4884 laser, a Coherent Cube 640 laser and a Coherent Cube 4057 laser and is capable of detecting 13 parameters. It is routinely used for 2-way and 4-way sorts at speeds of up to 20,000 cells/second as well as single cell deposition sorts. In addition to GFP, the most commonly used fluorochromes are FITC, PE, APC, PI, PE-TXRD, PE-CY5, APC-CY7 and Brillant Violet dyes. The MoFlo is also equipped with an Aerosol Evacuation Unit6 which will guarantee that no aerosols or particulates will escape from the sorting chamber. This will allow for safe sorting of human samples.
A BD Biosciences FACSAria IIIu2,5 desktop cell sorter is also available for cell sorting. This instrument has no counterparts with respect to capabilities and ease of use. It utilizes 488 nm, 640 nm, 561 nm and 407 nm air-cooled lasers with advanced optics to detect up to 18 independent signals. The 488 nm laser can be used to detect common fluorochromes including GFP, FITC and PerCP-Cy5.5. The 561 nm laser can excite dyes like PE, PI, and PE-Cy5, PE-Cy5.5 and PE-Cy7. The 633 nm laser is used to detect APC, APC-Cy7 and APC-Cy5.5. These fluorochromes are most commonly used by our investigators who sort hematopoietic progenitors or B cell subsets from tonsils. Finally, the 407 nm laser is appropriate for FRET experiments, exciting Quantum Dots, Brilliant Violet Dyes and for using Hoechst 33342 to study Side Population. The FACSAria also has an Aerosol Containment Device to ensure that any aerosolized particles are contained properly and making it safer to run human tissue samples.
The BD LSRII3 analyzer is equipped with 405 nm, 488 nm, 561 nm and 640 nm lasers. This digital instrument is a good companion to the FACSAria and is capable of detecting up to 19 parameters. Three fluorescent parameters from the 488 nm, three from the 640 nm, five from the 561 nm and six from the 405 nm.
The BD FacsCelesta8 analyzer is equipped with a 405 nm, 488 nm and 640 nm lasers and can detect 14 parameters. This digital instrument is capable of detecting 4 fluorescent parameters from the 488 nm laser, 5 parameters from the 405 nm laser and 3 parameters from the 640 nm laser.
The Cytek Aurora9,10 is a full spectra analysis flow cytometer. It has five lasers: 355nm, 405nm, 488nm, 561nm and 640nm. It can detect three scatter channels and 64 fluorescent channels. The Aurora can also measure the volume of acquired samples.
To assist researchers with analysis, the Flow Lab has a MacMini with SPICE and FlowJo and a PC with Diva, and FlowJo. Users can also inquire about signing up for OMRF’s FlowJo site license. We provide a transfer file on the network and have CDs and DVDs available to make copies of data files. We make routine backups of the data, but all users should frequently back-up their own data as well.
1The MoFlo was upgraded to a MoFlo XDP in 2012 from funds from an OCASCR grant. New PMTs, digital processors, computers and a solid state 640nm (Red) laser were installed, allowing better data resolution and performance.
2The Aria I was upgraded to an Aria IIu in 2012 from funds from an OCASCR grant. Fluidics were upgraded to a more stable and functional system, allowing for more reliable day to day performance.
3The LSR II was upgraded to a 4 laser system in 2010 from funds from an OCASCR grant. New 405nm (Violet) and 561nm (Green) lasers were installed and the 640nm (Red) laser was replaced. We expanded our capabilities from 12 to 17 fluorescent parameters.
4The MoFlo replaced the Enterprise II laser with a solid state Sapphire 488 laser with funds from an OCASCR grant in 2014.
5The FacsAriaIIu was upgraded to the FacsAriaIIIu with the addition of a Cube 561nm laser from funds from an OCASCR grant in 2015.
6The MoFlo was upgraded with an Aerosol Evacuation Unit with funds from an OCASCR grant in 2015.
7The MoFlo was upgraded with the Coherent Cube 405 laser with supplemental funds from an OCASCR grant in 2016.
8The FacsCelesta was purchased from funds from COBRE funds in 2016.
9The first Aurora was purchased with an S10 grant from NIH in 2020.
10The second Aurora was purchased with grants from OCASCR and PHF in 2022.
Standard Operating Procedures
All users must read through the appropriate SOP for the instrument that they are using and abide by the safety protocols at all times.
Contact


Gnotobiotic Mouse Core
Gnotobiotic (germfree, defined flora) rodents have become powerful tools to advance our understanding of the host–microbiome relationship. The maintenance and, ultimately, the monitoring of gnotobiotic rodents is a critical, labor-intensive and costly process requiring specialized skills and equipment.
The necessary gnotobiology techniques and equipment were developed more than 60 years ago. However, recently, the demand for gnotobiotic animals has rapidly expanded as the significant role of the microbiome in health and disease has become a major focus of research across multiple disciplines.
OMRF’s gnotobiotic mouse core provides centralized germ-free and gnotobiotic mouse services that include large flexible film isolators for breeding and small experimental isolators for a variety of in vivo studies utilizing germ-free mice. A specialized rack of sealed positive-pressure HEPA-filtered cages allows for microbiome control at the single-cage level, which is ideal for many applications. The core provides rederivation services for generating customized germ-free and gnotobiotic mouse strains, allowing for the study of the interaction of a gene of interest with the microbiome. The core provides technical support to investigators for numerous experimental procedures.
Services
- Caesarean rederivation — to create germ-free mice from existing SPF mouse lines
- Isolator setup and maintenance for dedicated projects
- Experimental study support
- Technical procedures
Contact

Jennie Criley, D.V.M.
Human Antibody Core Facility
The mission of the Human Antibody Core Facility at OMRF is to:
- Successfully collaborate with OMRF, OUHSC and external investigators to define temporal human immune responses after vaccination and to generate human monoclonal antibodies to supplement their research.
- Develop novel methods to produce human monoclonal antibodies after acute infection and other immune events.
- Investigate new technologies for the characterization of human monoclonal antibodies produced by the core.
The Human Antibody Core Facility is one of the few laboratories in the world that produces fully-human, full-length, antigen-specific antibodies for use in studying human immune responses. The Core has achieved breakthroughs in antibody technology and has produced hundreds of high affinity protective antibodies to influenza, anthrax lethal toxin, and various S. pneumonia polysaccharides. The Core aims to support investigators by helping quantify the antibody secreting cell responses after vaccination and by generating human monoclonal antibodies to be characterized. Pathogen-specific human monoclonal antibodies are also available for licensing agreements and other forms of commercial development. The Core is expanding these services to aid additional clinical and basic science investigators inside and outside Oklahoma.
Services & Pricing
Pricing is done on a project-specific basis, and an Agreement for Services will be provided once the price for the requested services has been determined.
- 20 Antibodies from ASCs After Vaccination (HAC does sort)
- 20 Antibodies from ASCs After Vaccination (customer does sort)
- Cloning of Antibodies (mouse or human) Into Expression Vector
- Expression of Antibodies (human or humanized)
- 4-plate transfection (50-500 micrograms of product)
- 20-plate transfection (250-2500 micrograms of product)
- 40-plate transfection (500-5000 micrograms of product)
- 80-plate transfection (1-10 milligrams of product)
- Elisa (1-4 plates, customer supplies antigen)
- Hybridoma Sequencing
- Site-specific Mutagenesis or Reversion of Antibody or Germline
- Non-antibody Protein Express (in HEK293 or BL21)
Contact


Myositis Testing
Some types of autoimmune disease attack the muscles of the body. The “myositis-specific antibodies” (MSA) can assist in the diagnosis of polymyositis and dermatomyositis in those patients who have the diseases. About 50% of patients with polymyositis or dermatomyositis have specific MSA or myositis associated antibodies (MAA). MSA are almost never found in patients without myositis, even if they have other muscle diseases of autoimmune diseases. This means that when the physician’s examination and initial testing suggest the possibility of polymyositis or dermatomyositis, a positive test for an MSA can be strong supporting evidence for the diagnosis.
For a long time the testing for these antibodies was only available in research studies, but it is now possible to obtain this testing clinically. The Myositis Profile* performed at the OMRF Clinical Immunology Laboratory includes tests for 12 of the MSAs and MAAs. Additional antibodies may be detectable using this profile. Dr. Ira Targoff is the consultant to the Clinical Immunology Laboratory regarding this testing. As with the Reichlin Profile for lupus, patients with a particular antibody may have distinctive clinical pictures with certain associated problems. Identifying certain antibodies can help characterize the condition more fully. Research continues in our lab, and others around the world, to determine the nature of the relationship between the various MSAs and polymyositis and dermatomyositis.
Contact
To contact the OMRF Myositis Testing Laboratory staff, call 405-271-7397.
Federal accreditation regulations prohibit laboratory personnel from providing patient information or results over the phone or via email. Patients, please contact your health care provider for questions concerning your results.

Research Computing Services
OMRF’s Research Computing provides a High-Performance Computing (HPC) cluster for all OMRF researchers to conduct their data science in a fast and efficient manner. This Linux-based HPC cluster is housed at OMRF and is regularly updated to provide for changing computing needs. The cluster currently includes both high-memory nodes and GPU nodes. All systems are connected to high performance and large capacity storage.
Nanopore PCR and Plasmid Sequencing
Utilizing Oxford Nanopore Technology sequencing platforms we have the ability to sequence entire plasmids and amplicons (>600 bp) without sequencing primers or PCR. Runs are performed Monday, Wednesday, and Friday. For technical questions and assistance, contact Graham Wiley.
- DNA must be 30-50 ng/ul ≥ 10 ul and quantified using a fluorometric method (i.e. Qubit)
- Files returned will include a .fasta sequence file and an .html summary file containing a complete annotation of each plasmid, along with read length and quality histograms
- Sample Submission Cutoffs:
Samples for Monday runs must be submitted by 5 pm the previous Friday.
Samples for Wednesday runs must be submitted by 5 pm the previous Tuesday.
Samples for Friday runs must be submitted by 5 pm the previous Thursday. - Sample drop-off points are the refrigerators in rooms E324 and C102.
- Accessing your results: Select the “View My Requests” tab in iLab. The file is under your sample manifest.

Collaborations
The Oklahoma ACE Program has successfully forged numerous collaborations across scientific disciplines, across medical specialties, and between basic and clinical scientists in the search for effective treatments for autoimmune diseases.
Clinical studies:
- SRA01
- ASJ02 – Studies Baminercept in primary Sjögren’s syndrome; led by Judith A. James, MD, PhD
- ALE06 – Investigates the withdrawal of MMF in suppressed SLE; led by Eliza Chakravarty, MD
- ARA08 – A collaboration with Kevin Deane, MD, PhD and Michael Holers, MD of the University of Colorado to prevent the onset of clinically-apparent rheumatoid arthritis (also known as StopRA)
- ALE09
- AMS04
Mechanistic studies:
- ASJ02 – A collaboration with Duke University that studied the mechanisms of B cell responses in autoimmune disease
- ALE06 – A collaboration with Duke University to determine the mechanisms of B cell responses in autoimmune disease; Directed by Judith A. James, MD, PhD and Joel Guthridge, PhD
- ARA08
- ALE09
Education
The Oklahoma ACE co-sponsors a weekly Autoimmunity and Clinical Immunology Research Forum, partners with OMRF to bring in outside speakers for the OMRF Research Forum, and co-hosts a journal club and clinical conference. At the Autoimmunity Research Forum, members present works-in-progress and receive feedback in a collegial, yet scientifically critical, interdisciplinary setting. This is a critical tool for allowing our members to strengthen their hypotheses, experimental plans, and conclusions before external peer review, and for helping our junior members develop strong presentation skills. The Arthritis and Clinical Immunology Journal Club provides an opportunity for members to delve into important developments in the literature. Finally, the Rheumatology Clinical Conference helps connect basic and translational researchers with clinical investigators, providing our members with a strong clinical perspective on their research.
Contact
Judith A. James, M.D., Ph.D., is the Director of the Oklahoma ACE program and also serves as the Principal Investigator of the Administrative Core. The goals of the Administrative Core are:
- To provide central management for Oklahoma ACE components and activities
- To facilitate interactions of the Oklahoma ACE with other members of the ACE Network and the NIH ACE Program
- To provide project management support progress of clinical and collaborative projects
- To promote data-sharing strategies
Dr. James also serves as leader of the Collaborative Project, along with Joel Guthridge, Ph.D. This project focuses on evaluating and refining molecular signatures and assessing the roles of monocytes and B cells in ACE cohorts and ACE clinical trials to aid treatment selection and clinical trial design.

Judith James, M.D., Ph.D.
Autoimmunity Prevention Center
Systemic lupus erythematosus (SLE) can damage nearly any organ in the body, and this organ damage often begins even before patients are diagnosed. Therefore, the OMRF Autoimmunity Prevention Center focuses on understanding the processes that precede and lead up to lupus. This research is important for identifying patients at high risk of SLE and developing therapeutics that safely interrupt the disease process to prevent organ damage.
Research in this area has been hindered by the challenge of identifying and collecting samples from patients who will go on to develop SLE. To overcome this obstacle, we have assembled five cohorts that span the spectrum of preclinical autoimmunity from genetic risk to SLE classification (Figure 1). The OMRF Autoimmunity Prevention Center is using these resources to delineate the progression of immune dysregulation leading up to SLE classification, explore mechanisms of disease transition that could be targeted by preventive interventions, and validate algorithms for identifying individuals at high risk of SLE.
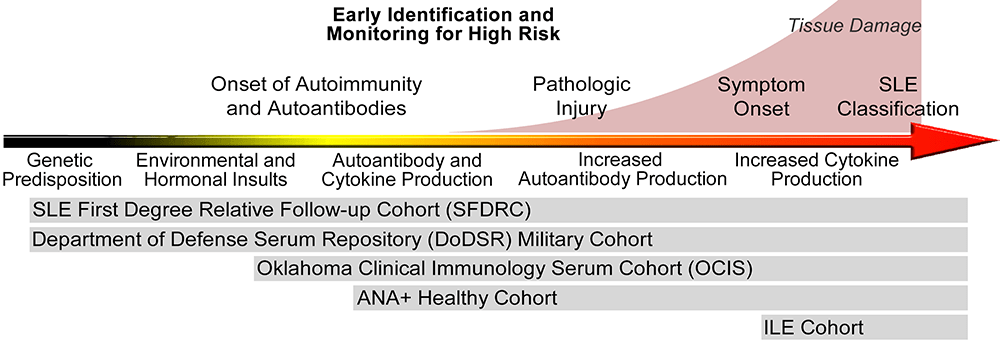
We actively collaborate with other Autoimmunity Prevention Centers across the nation. These collaborations have led to the first ever lupus prevention trial, called Starting anti-Malarials in ILE (SMILE: NCT02603146), as well as the first RA prevention trial in the US (StopRA: NCT02603146).
The OMRF Autoimmunity Prevention Center was established in 2012 with funding through the NIAID Cooperative Study Group for Autoimmune Disease Prevention (U01AI101934). This group is a collaborative network of investigators focused on preventing autoimmune diseases before clinical onset.

Contact CBDS

Courtney Montgomery, Ph.D.
Read Bio
Courtney Montgomery, Ph.D., is the director of the CBDS and a professor in the Genes & Human Disease Research Program. She has over a decade of experience in directing Biostatistics and Bioinformatics Cores for Centers of Research Translation (CRT) and Centers of Biomedical Research Excellence (COBRE) grants including managing a team of skilled biostatisticians, bioinformaticians, statistical geneticists, and data administrators. She was named the founding Director of the CBDS beginning May 2023. Her extensive training and experience in biomedical data sciences includes statistics, genetic epidemiology, and bioinformatics. As a member of the Human Genetic Analysis Resource at Case Western Reserve University, she spent several years working on the development and refinement of methodology for statistical analysis of genetic data, with a special emphasis on highly correlated hierarchical data modeling methods. She has been the PI of multiple NIH and foundation grants and leads a team of statisticians and programmers that is involved in a variety of clinical, translational, and basic science projects. Her specific research interests include the genetics of sarcoidosis and other autoimmune disorders, as well as the development and application of novel statistical and bioinformatics approaches to large-scale, high-dimension data.

Christopher Bottoms, Ph.D.
Read Bio
Christopher Bottoms, Ph.D., is a Senior Bioinformatics Trainer/Analyst with over two decades’ experience using GNU/Linux, Bash, and programming languages, thirteen years’ experience with git, twelve years’ experience using HPC clusters, and a decade’s experience using Slurm. He has a strong background in biology, genetics, and protein structures and previously served as the senior Programmer/Analyst at the University of Missouri Bioinformatics Analytics Core. His past work included numerous plant- and animal-based model systems and multiple sequencing and analytical platforms.

Nathan Pezant, MS
Read Bio
Nathan Pezant, MS, is a Senior Data Analyst in the CBDS. He has Master’s degrees in both Applied Mathematics and Statistics. His past work focused on the bias and accuracy of gene expression classification models under various resampling schemes. Since joining OMRF in 2016, Mr. Pezant has provided analyses and consultations to many researchers at OMRF and surrounding research institutions. Mr. Pezant has experience with many types of genetic and clinical data as well as programming and scripting languages.

Gideon Hallum, MS
Read Bio
Gideon Hallum, MS, is a Bioinformatics Project Manager in the CBDS. He serves as liaison between CBDS staff and other OMRF personnel including scheduling, project tracking, and other administrative reports. He has a Master’s Degree in Biostatistics and experience in survival analysis, sample size calculation, and cancer research and is versed in multiple programming languages and data types.
Centers of Biomedical Research Excellence (COBRE)
With grants from the National Institute of General Medical Sciences, OMRF has established five centers of excellence. Centers of Biomedical Research Excellence (COBRE) grants help build necessary infrastructure to enhance OMRF’s research capacity and competitiveness for National Institutes of Health grants. In addition, they help promote collaboration among scientists with complementary backgrounds and expertise. COBRE support also provides a platform for mentoring relationships between well-established scientists and up-and-coming researchers within a discipline.
Active
Center for Genomic Regulation
Grant #P20GM156711
Cellular Metabolism Research in Oklahoma
Grant # P20GM139763-01
Expanding Excellence in Developmental Biology in Oklahoma
Grant # P20GM103636
Complete
Interdisciplinary Research in Vascular Biology
Grant # P30GM114731
Science in a Culture of Mentoring
Grant # P30GM103510
Molecular Mechanisms and Genetics of Autoimmunity
Grant # P20GM103456
Molecular and Immunologic Analysis of the Pathobiology of Human Anthrax (U-19)
We have built and continue to advance our multidisciplinary approach to the study of the human immune response to Bacillus anthracis. We view B. anthracis as one of the most significant threats to our society as a bioterrorist agent, distinct from its use as a biological weapon. B. anthracis is alarming as a weapon of terror for several reasons: 1) the spore is long-lived, is simple to make, does not require complex and expensive equipment, and is stable in harsh environments; 2) the vaccine is of questionable efficacy, has a cumbersome injection schedule, and is not provided to civilians; 3) mortality remains high even with effective antibiotic therapy ; 4) even a small number of deaths from a rare and thus exotic disease is sufficient to cause terror in our population. Indeed, this is exactly what happened after the US Postal Service Center attacks in 2001.
The group we assembled is productive, highly interactive and has a formidable background in immunology as well as expertise in Bacillus anthracis. Our group includes faculty, fellows, technicians, veterinary staff and other support staff at four different institutions: The Oklahoma Medical Research Foundation, The University of Oklahoma, The University of Chicago, The University of Cincinnati, and Boston University. Our efforts consist of five Scientific Projects, three Technical Projects and two Core Facilities (along with Administrative and Educational cores). We attack the problem from every direction: powerful experimental models largely developed in house, a novel approach to vaccine genetics and vaccine efficacy, several host-response interactions, studies of the exotoxins, and two novel and exciting therapeutic approaches.
Our efforts are on-going over seven years and we have made substantive advances. Our ultimate goal is to eliminate anthrax from the bioterrorist arsenal.
Oklahoma Nathan Shock Center for Excellence in the Basic Biology of Aging
The Nathan Shock Center for Excellence in the Basic Biology of Aging was established in 2015 with a grant from the National Institutes of Health. It is one of only six Shock Centers nationwide.
Through the center, researchers from the University of Oklahoma Health Sciences Center, OMRF and the Veterans Administration Medical Center work together to address the ways aging impacts disease and changes that occur in aging that predispose people to disease.
The Shock Center has three key functions:
- To develop a statewide presence as a leader in geroscience, where scientists can collaborate with colleagues at a wide range of institutions;
- Provide funding to support the development of aging-targeted research projects; and
- Provide unique services to researchers that are unavailable from other sources in Oklahoma
Shock Center staff also serve as mentors for junior investigators, especially at institutions with little aging research. They also provide leadership by directing younger researchers and allowing them access to core facilities to further their research projects.
The Shock Center grant also has established the Oklahoma Geroscience Consortium, which brings together aging-focused scientists at OUHSC, OMRF and VAMC, as well as Oklahoma State University and the OU campus in Norman.
Oklahoma Rheumatic Disease Research Cores Center (ORDRCC)
Who we are
Over 200 researchers spanning the basic, clinical and analytical sciences
What we do
Advancing prediction, prevention and precision therapy for patients with rheumatic diseases
Why we care
Patients with rheumatic diseases need better tests and personalized treatments so they can live healthier, longer lives
Extensive cohort collections
Our CAP-accredited biorepository manages some of the largest existing collections of SLE, Sjogren’s, RA, and osteoarthritis samples
Cutting-edge methodologies
The Human Phenotyping Core offers single-cell technologies, including CyTOF, Spatial Transcriptomics, and scRNASeq
Clinical research expertise
The Clinical Core assists with IRB approvals, study recruitment, and clinical research visits
Oklahoma Shared Clinical and Translational Resources (OSCTR)
Through a partnership between the University of Oklahoma Health Sciences Center, OMRF and other partners statewide, the Oklahoma Shared Clinical and Translational Resources (OSCTR) stimulates research that meets the health needs of underserved and underrepresented Oklahomans. As one of just nine such centers in the United States, The OSCTR is creating a more competitive landscape for clinical and translational research in Oklahoma by:
- Developing infrastructure for clinical and translational research
- Funding promising new lines of research and junior investigators through pilot grant funding
- Building a pool of well-trained and committed clinical and translational investigators through education, mentoring and career development
- Facilitating rigorous and interpretable studies through consultation, collaboration, and training on biostatistics, epidemiology and research design
- Engaging the community to identify research priorities, refine innovations and spread effective innovations to clinical practice
- Providing access to cost-effective clinical resources for research, including large, well-characterized biospecimen collections, regulatory assistance, subject recruitment assistance and staffed facilities for clinical research visits
- Fostering respectful collaborations and investigations within tribal communities and other underserved populations
- Promoting effective progress through well-designed program evaluations
- Implementing strategic leadership, central oversight and management of personnel and resources devoted to the OSCTR
A $20.3 million grant from the National Institutes of Health (U54GM104938) supports the OSCTR through an Institutional Development Award (IDeA). The IDeA program fosters health-related research in states with historically low NIH grant funding success rates, enabling them to become nationally competitive.
Oklahoma Sjögren’s Syndrome Center of Research Translation (OSSCORT)
The Oklahoma Sjögren’s Syndrome Center of Research Translation (OSSCORT) has been developed with the goal of implementing the resources and administrative structure to facilitate the translation of laboratory-based research in Sjögren’s syndrome into relevant diagnostic, prognostic and therapeutic innovations in the field of autoimmune disease.
The OSSCORT has the unique opportunity to bring together a strong core of well-funded laboratory-based scientists and clinical researchers at the Oklahoma Medical Research Foundation, with a multidisciplinary network of investigators committed to Sjögren’s syndrome research from the University of Oklahoma Health Sciences Center and multiple U.S. and international sites.
The OSSCORT’s first phase consists of three projects:
- Project 1 addresses the genetics of SS by applying state-of-the-art genome wide association, fine-mapping and replication experiments, as well as genotypic delineation of sub-phenotypes.
- Project 2 has developed innovative approaches to reveal key features of T cell immune dysregulation in Sjögren’s
- Project 3 utilizes in vitro and a humanized mouse model of SS to test the hypothesis that B cells infiltrating the salivary glands of patients make autoantibodies that are in part responsible for the exocrine glandular dysfunction.
Three cores support the OSSCORT, which also has supported a highly productive Enrichment Program. The Cores that assist and facilitate the OSSCORT are an Administrative Core, a Clinical Core and a Data Analysis and Bioinformatics Core.
About Sjögren’s
Sjögren’s syndrome (SS) is a chronic, progressive autoimmune disorder characterized by lymphocytic infiltration of the exocrine glands that leads to severe oral and ocular dryness. SS is also a systemic disease that may include debilitating fatigue, lymphoma, neuropathies, pulmonary disease and arthralgias. The etiology of SS is poorly understood but clearly involves a complex genetic architecture influenced by environmental factors that lead to abnormal cellular and humoral immune responses. The difficulty in correctly diagnosing SS in addition to the scarcity of effective therapeutic options, often results in significant morbidity and irreversible damage to the exocrine glands.
Samuel Roberts Noble Cardiovascular Institute
The Samuel Roberts Noble Cardiovascular Institute was established in 2008 with a gift to OMRF from Ardmore’s Noble Foundation. For decades, the Noble Foundation and family have supported research at OMRF. The Institute is named in memory of the late Sam Noble, a long-time OMRF board member whose father and legendary oilman Lloyd Noble established the family’s foundation. Lloyd Noble ultimately lost his life to a heart attack in 1950.
The Noble Institute serves as home to OMRF’s Cardiovascular Biology Research Program, where scientists study blood and lymphatic vessel function, blood coagulation, inflammation and sepsis, a deadly form of blood poisoning. Research in this program also yields new insights on a variety of human diseases including heart attack, stroke, cancer and inflammatory bowel disease.
Projects
Lijun Xia, M.D., Ph.D.
Xia’s work focuses on O-glycans, a form of sugar that the body produces and that comprises nearly 80 percent of a thick mucous layer inside the colon and the gastrointestinal tract. These sugars produced by the body play an important role in the development of colitis, Crohn’s disease and colon cancer.
Ahamed joined OMRF in 2015 from The Rockefeller University, and his research centers on how blood clotting leads to the release and activation of a multifunctional protein that contributes to cancer, autoimmune disorders and cardiovascular disease.
In the lab, Griffin use genetics, molecular biology, biochemistry and developmental biology techniques to study blood vessel and lymphatic system development in embryos and adults. She also focuses on programmed cell death and the processes that can lead to atherosclerosis and aneurisms.
In his lab, Olson studies fibrosis, the creation of scar tissue in disease. Fibrosis can cause heart, kidney and liver failure, and it can also be an early event in the disease process leading to diabetes.
The lymphatic system is a network of vessels and lymph nodes that spans the entire body, absorbing tissue fluids and returning them to the blood stream. Srinivasan studies the defects that can lead to lymphedema, a dramatic and painful swelling in the limbs that can often lead to infections.
Trans-Agency Consortium for Trauma-Induced Coagulopathy (TACTIC)
Led by OMRF scientist Charles Esmon, Ph.D., TACTIC brings together leaders in the field to study coagulopathy, a deadly bleeding syndrome resulting from traumatic injury. The TACTIC study is a cooperative effort funded by the National Heart, Lung and Blood Institute that establishes a unique collaboration between the National Institutes of Health and the Department of Defense. This unique initiative integrates laboratory, clinical and early translational, hypothesis-driven research by leading investigators across the country and enables the basic science investigative units to explore clinical specimens obtained from the DoD centers.
Additional institutions involved in the research funded by the TACTIC grant include Massachusetts Institute of Technology, Mayo Clinic, Scripps Research Institute, University of California-San Francisco, University of Illinois and University of Pennsylvania. DoD-supported institutions participating in the clinical component of the TACTIC grant include University of Colorado, University of Pittsburgh, and Virginia Commonwealth University.
About Coagulopathy
Trauma is the major cause of death in people under age 34 and the third leading cause of mortality in the U.S., with uncontrollable hemorrhage representing the major cause of preventable deaths, according to the NIH. Each year there are nearly 50 million injuries in the U.S. that result in 170,000 deaths.
Little is known about the biological phenomena that lead to coagulopathy. When a people sustain a traumatic injury, whether on the battlefield or in a car accident, they typically suffer serious physical damage. Doctors treat the injuries and, if all goes well, the patient gradually heals. However, some patients, regardless of proper treatment, can suddenly suffer from uncontrolled bleeding and die. It is believed that the shock from the trauma induces a “storm” of coagulation and inflammatory problems that prevents blood from clotting.
In this comprehensive initiative, TACTIC researchers and clinicians are combining their efforts to identify the steps that trigger coagulopathy and new ways to treat it.

Comparative Medicine
The department of Comparative Medicine manages the OMRF animal care program’s three facilities. It has been accredited by AAALAC International since 1971 and is a member of the Vivarium Operational Excellence Network.
The department manages an average of approximately 7,800 cages of mice and 1,200 tanks of fish. Our cage rates rank among the lowest in the country, and we are capable of housing and maintaining rats, frogs, other rodents and other species as needed. For all rodents, the quality of housing is barrier-level with sterile caging and bedding, irradiated feed and chlorinated RO water. Comparative Medicine offers the capability of ABSL-2 containment for different projects and can accommodate ABSL-3 housing in collaboration with neighboring institutions.
The attending veterinarian is involved in protocol development, provides day-to-day (including weekends) clinical and operational oversight, and conducts post-approval monitoring of animals at cage-side. In addition to offering barrier-quality services, Comparative Medicine provides breeding and colony management, tail snips, injections, blood and sample collection, euthanasia, treatments, import/export, osmotic pump implants and customized services as needed. Staff also conducts no-cost training for lab personnel in areas ranging from basic animal handling to anesthesia and invasive procedures.
Available imaging and in vivo modalities include:
- Micro–CT
- Dexa scanner
- Echo MRI
- Visual Sonics (Vevo 2100) ultrasound
- IVIS Lumina in vivo imaging
- Radsource RS 2000 small animal irradiator
- Rodent treadmill and metabolic caging
Contact

Jennie Criley, D.V.M.
Room B-120
825 NE 13th St.
Oklahoma City, OK 73104
Facility Services
Facility services provides professional support for the design, construction, operation and maintenance for all OMRF facilities, which consist of more than 500,000 square feet of laboratory, administrative, support and clinical space.
With a staff of 12 licensed and certified technicians, facility services maintains, operates and repairs critical building systems (air conditioning, electrical, gas, heat, plumbing, ventilation, water) and all campus facilities. The department performs an average of more than 3,000 preventive and repair jobs a year, and it uses a comprehensive software system to monitor more than 3,000 air handlers, chill water pumps, phoenix valves and humidity and temperature points throughout OMRF.
Institutional Biosafety Committee
The Institutional Biosafety Committee (IBC) reviews, approves and oversees research involving the use of recombinant or synthetic DNA/RNA and other biohazards. In accordance with the NIH Office of Science Policy update, Implementation Update: Promoting Maximal Transparency Under the NIH Guidelines for Research Involving Recombinant or Synthetic Nucleic Acid Molecules, for all OMRF IBC meetings taking place on or after June 1, 2025, the IBC will post approved meeting minutes here. IBC meeting minutes will be posted immediately after approval and after all appropriate and allowable redactions are made.
Meeting Minutes
June 2025 – no meeting held
July 2025 – no meeting held
August 2025 – no meeting held
September 2025 – no meeting held
October 2025 – minutes pending approval
November 2025 – no meeting held
December 2025 – no meeting held
January 2026 – meeting minutes
February 2026 – minutes pending approval
Contact
405-271-6670 ext. 32935
IBC@omrf.org
Grants & Contracts
The office of grants and contracts assists OMRF faculty in the pursuit of external funding opportunities, providing support services throughout the life (pre-award, post-award and closeout) of all sponsored projects. They also work to ensure compliance with applicable regulations and requirements.
Five full-time staff members provide the following services:
- Research administration
- Assistance in proposal preparation
- Proposal submission
- Project administration, including:
- Grant award set-up
- Execution of sub-contracts
- Budget monitoring and compliance
- Reporting
- Grants accounting
- Administrative training for grant and grant-related activities
Contact

Stephanie Mode
Information Technology
A team of IT professionals that includes three staff members dedicated to help desk support, six staff members in IT operations/infrastructure, and six application/web developers and business analysts.
IT Support
- Technical support for all OMRF owned workstations, desktop and laptop computers
- Software support for networked applications
IT Operations, Infrastructure, and Telecommunications
- Data storage and data transfer tools
- Networking
- Server hosting and support
- Technical consulting services
- VoIP phone and mobile phone support
Information Security
- Secure file sharing for collaboration, VPN, encryption, backups, end-point security, email filters, firewall protection, etc.
Application/Web Development and Business Analysis
- Application development and support
- Design and maintenance of departmental, investigator and lab web pages
Contact
Information Technology
405-271-7470
InformationTechnology@omrf.org






